Three-Electrode System: The Foundation of Electrochemical & Battery Research
Table of Contents
- What Is a Three-Electrode System?
- Components and Their Functions
- Why the Three-Electrode System Matters in Battery Research
- How the Three-Electrode System Works
- Practical Tips for Better Electrochemical Testing
- Performance Parameters (Table)
- Advanced Testing with ACEY Battery Lab Research Machines
- Data Analysis and Research Applications
- FAQs
- Partner with ACEY Intelligent
If you've ever wondered how researchers deeply analyze the performance of battery materials, the answer lies in the three-electrode system. Unlike everyday two-terminal batteries, the three-electrode setup is specifically designed for precise electrochemical control and measurement in laboratory environments.
It allows scientists to independently measure voltage and current, enabling accurate evaluation of electrode reactions-something that's vital for developing high-performance lithium-ion batteries and next-generation energy storage systems.
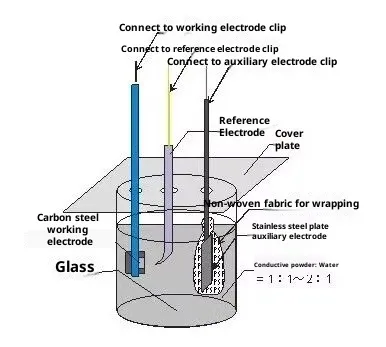
A typical three-electrode cell includes:
| Electrode Type | Function | Common Materials |
|---|---|---|
| Working Electrode (WE) | The main electrode where the reaction occurs | Glassy carbon, platinum, gold, or test battery electrodes |
| Counter Electrode (CE) | Balances the current and completes the circuit | Platinum wire, graphite rod |
| Reference Electrode (RE) | Provides a stable voltage reference | Ag/AgCl, saturated calomel electrode |
Each electrode plays a unique role-together, they allow precise potential control and current measurement, the two pillars of reliable electrochemical testing.
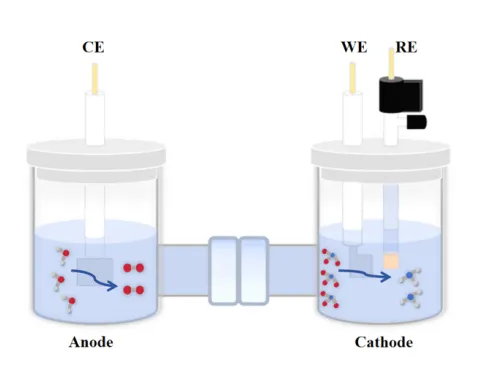
Early electrochemical studies used two-electrode setups, which often produced inaccurate data. The introduction of a reference electrode in the three-electrode system revolutionized battery research by allowing scientists to:
- Precisely control electrode potentials, avoiding current interference.
- Minimize voltage errors caused by solution resistance or electrode polarization.
- Study reaction mechanisms in complex battery materials more clearly.
This configuration is now a standard tool in labs studying lithium-ion batteries, fuel cells, and supercapacitors.
The system operates through two interconnected circuits:
- Potential Circuit: Measures the voltage difference between the working and reference electrodes.
- Current Circuit: Controls current flow between the working and counter electrodes.
This dual-circuit setup is the basis of techniques like:
- Cyclic Voltammetry (CV) – for studying redox behavior
- Electrochemical Impedance Spectroscopy (EIS) – for understanding internal resistance
- GITT/PITT – for analyzing diffusion and kinetics
Together, they provide a complete electrochemical picture of how materials behave during battery charge and discharge cycles.
To achieve accurate, reproducible results, researchers should:
- Place the reference electrode close to the working electrode to minimize resistance.
- Use a large counter electrode to prevent polarization.
- Clean and standardize the working electrode surface.
- Apply iR compensation carefully to reduce measurement error.
- Select the right cell design (beaker cell or coin/pouch-cell adaptor) based on experiment type.
| Parameter | Example Value | Description |
|---|---|---|
| Voltage Control Accuracy | ±100 μV at 4.5 V setpoint | Ensures potential stability |
| Current Measurement Accuracy | ±0.01% (100 mA ± 5 μA) | Reliable for micro-scale current detection |
| Noise Level | < 0.01% RMS | Low interference for clear signal |
| Frequency Range (EIS) | 0.01 Hz – 100 kHz | Covers wide electrochemical responses |
| Supported Techniques | CV, EIS, GITT, PITT | Full electrochemical analysis suite |
These parameters are crucial when using ACEY's battery lab research machines, ensuring stability and repeatability in all electrochemical tests.
At Xiamen Acey Intelligent Equipment Co., Ltd., we design and manufacture high-precision battery lab research machines that support all major electrochemical testing methods.
Our systems feature:
- Ultra-low noise circuitry for clean, reliable signals.
- Wide frequency response for detailed EIS analysis.
- Nanoamp-level current detection for micro-scale reactions.
- Intelligent data software for real-time visualization and analysis.
These tools empower researchers to explore new materials, improve SEI formation control, and enhance lithium-ion battery performance with laboratory precision.
ACEY's electrochemical analyzers are integrated with advanced software capable of multi-dimensional data visualization, curve fitting, and automated parameter extraction.
This enables researchers to:
- Understand charge-transfer kinetics.
- Quantify diffusion coefficients in electrode materials.
- Evaluate SEI film impedance through EIS.
- Optimize battery design for energy density and cycle life.
Q1: What's the difference between a two-electrode and three-electrode system?
A two-electrode setup can't separate potential control from current measurement, while a three-electrode system provides independent control, making it much more accurate for research.
Q2: Why is a reference electrode necessary?
It provides a stable potential baseline, allowing precise measurement of the working electrode potential without being affected by current flow.
Q3: Can I use ACEY analyzers for lithium-ion battery testing?
Absolutely. ACEY's electrochemical workstations and battery lab research machines are designed for battery materials, electrodes, and full-cell studies.
Q4: What techniques can the ACEY analyzer perform?
Our analyzers support CV, EIS, GITT, PITT, and chronoamperometry-all essential for advanced electrochemical research.
Whether you're a research lab, university, or battery manufacturer, ACEY Intelligent is your trusted partner for advanced electrochemical testing equipment.
We're currently seeking international agents, brand ODM partners, and distributors to expand our global reach.
👉 Join us today and bring world-class battery lab research machines to your region.
📩 Contact us at www.aceyintelligent.com to learn more.